The pharmaceutical industry is one of the most highly regulated sectors in the world. Before any medicine reaches patients, it must undergo rigorous regulatory evaluation to ensure its safety, efficacy, and quality. This vital responsibility is managed by the field of Drug Regulatory Affairs (RA).
Regulatory Affairs serves as the backbone of the pharmaceutical industry by ensuring that pharmaceutical products comply with national and international regulatory standards. It is a specialized professional field focused on the preparation, submission, and lifecycle management of documentation required to obtain and maintain marketing authorization for medicinal products.
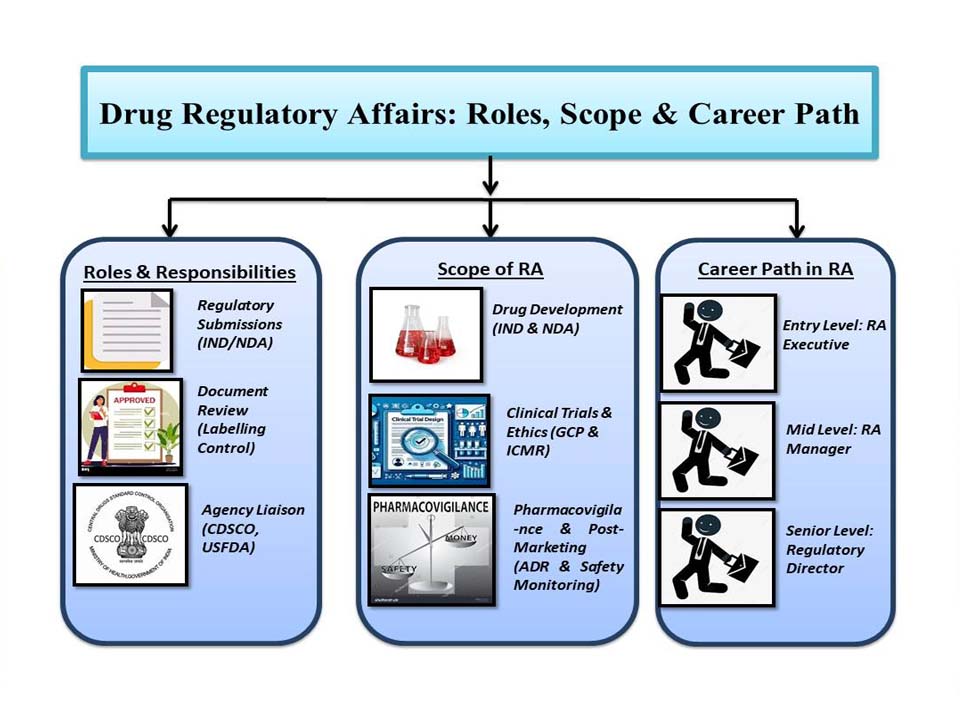
Key Responsibilities of RA Professionals
- Prepare regulatory submissions for product approvals and renewals.
- Review labelling, packaging, and technical documentation.
- Liaise with regulatory agencies like CDSCO, USFDA, or EMA.
- Track and interpret updates to global regulatory frameworks.
- Maintain product registration databases and compliance records.
Scope of Drug Regulatory Affairs
- Drug Development & Regulatory Approvals
Regulatory professionals are involved from the early stages of drug development through to product launch and post-marketing activities. They are responsible for preparing and submitting regulatory drafts such as Investigational New Drug (IND) applications and New Drug Applications (NDAs), as well as ensuring continuous compliance with regulatory requirements throughout the product lifecycle.
- Clinical Trials & Ethics
RA professionals work closely with clinical research teams as well as regulatory agencies to make the drug development process faster and more efficient. They ensure that every step is well above approval levels, allowing for a smoother and quicker launch to market. They maintain regulations with Schedule Y, GCP, and ICMR guidelines and support ethical and effective clinical trial management.
- Medical Devices & Biotechnology
RA professionals confirm that the medical devices are in line with CE marking and new Indian regulations for medical devices. With the growth of biotechnology and biologics, the role of RA has expanded to include areas like vaccines, biosimilars, and advanced therapies.
- Pharmacovigilance & Post-Marketing Surveillance
After a drug is approved, RA professionals continue to keep track of its safety by reporting adverse drug reactions (ADRs). They also conduct audits to make sure production still follows all the approved quality standards. To maintain scientific validity, approved labelling compliance, review marketing collateral and product claims.
- Regulatory Affairs in R&D and Product Lifecycle
RA professionals are now seen as strategic partners in product development. They help manage legal and technical complexities early in product development. They communicate with authorities, help bring products to market faster, and take action for any problems that arise.